Altering a subscript would change the molecule itself. The subscript, however, cannot be changed. Changing the coefficient changes the total number of that molecule. The coefficient is the part that can be changed and added when balancing an equation. 
The 2 is multiplied by the subscript of hydrogen which is 3, yielding a total number of hydrogen atoms equal to 6. For example, in the second picture, the coefficient for ammonia (NH3) on the products side is 2. There are many sample equations in this chemical equation balance calculator so that you can practice and balance equations. If you are using balancing chemical equations calculator with steps, you'll know how easy it is to operate. If an atom has a subscript, the coefficient and the subscript multiply to yield the total amount of that atom in the molecule. This balance chemical equations calculator helps to balance chemical equations quickly. For example, in the first picture the coefficient for the second term indicates that 3 molecules of H2 are present, and the subscript of the first term signifies that 2 atoms of nitrogen (N) are present per molecule of N2.Īdding a coefficient in front of a molecule multiplies all atoms within that molecule by the number of the coefficient. 
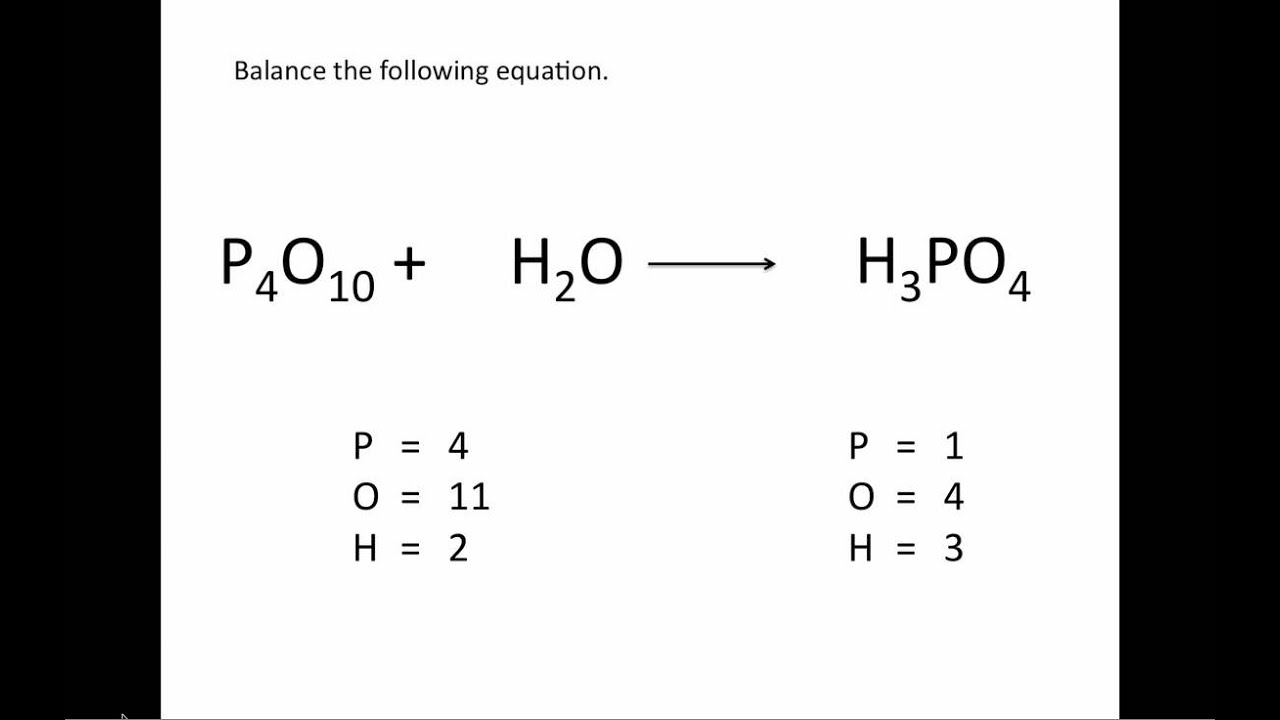
The subscript of an atom indicates the amount of that atom in the molecule. There is only one atom of carbon on the left-hand side, but six on the right-hand side. Here, both carbon and hydrogen fit this requirement. In a molecule, the coefficient denotes the amount of that molecule present. The first step to balancing chemical equations is to focus on elements that only appear once on each side of the equation. The coefficient is placed in front of a molecule, while the subscript follows certain atoms as shown in the first picture. When approaching a chemical equation, it is important that you understand the difference between coefficients and subscripts. The examples will only use whole numbers and will not discuss equations that involve complex ions, which is a molecule that has a charge. The instructions will examine balancing simple equations that contain 2 molecules for reactants and for products. In simplified terms, the law states that there must be an equal number of atoms of each element in the reactants as in the products. It is important to balance chemical equations in order to follow the Law of the Conservation of Mass. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. Abbreviations can be found in a periodic table of elements. Step 4: Substitute Coefficients and Verify Result. Abbreviations of element names are used to facilitate the equations. Generally, they follow the format of reactants to products, where "reactants" are the starting materials of your reaction and "products" are the end result.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
